Organic Chemistry: Building Blocks of Life and Innovation
Organic Chemistry: Building Blocks of Life and Innovation
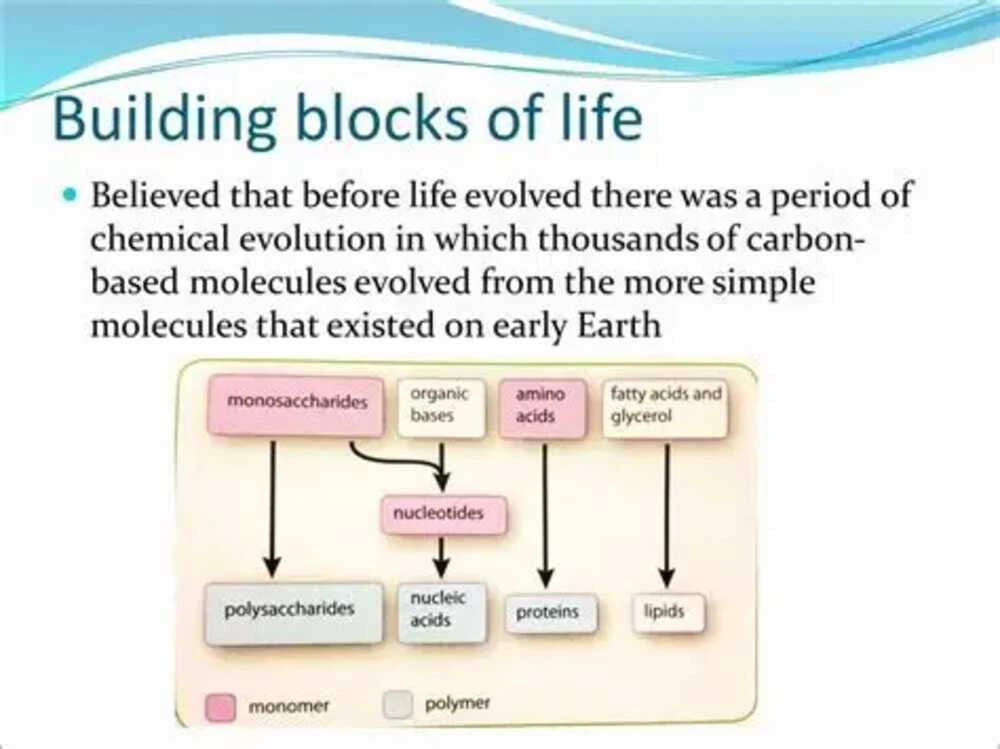
Why Organic Chemistry Matters More Than Ever
Organic Chemistry sits at the center of modern science. It is the study of carbon-based compounds, the molecules that form living organisms, medicines, fuels, plastics, food additives, cosmetics, and advanced materials. For students entering chemistry, biology, medicine, or engineering, understanding Organic Chemistry is not just another academic requirement. It is a practical foundation for solving real-world problems.
From the DNA in our cells to the polymers in smartphones, Organic Chemistry explains how molecular structures influence function. This makes it one of the most useful branches of science for innovation. Industries across pharmaceuticals, agriculture, biotechnology, and manufacturing rely heavily on organic compounds to create safer, more efficient, and more sustainable products.
For students, learning Organic Chemistry can feel intimidating at first. Complex structures, reaction mechanisms, and naming conventions often seem overwhelming. But once the core principles are understood, the subject becomes less about memorization and more about pattern recognition and logic.
The Carbon Advantage in Organic Chemistry
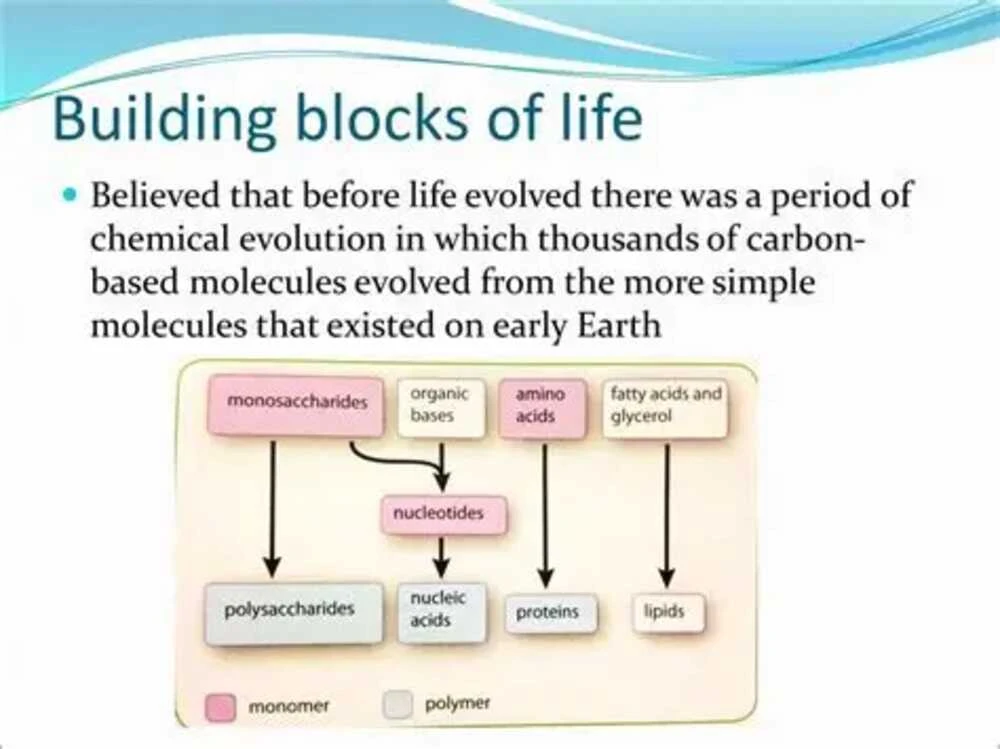
At the heart of Organic Chemistry is carbon, a uniquely versatile element. Carbon has four valence electrons, allowing it to form four stable covalent bonds. This flexibility enables carbon atoms to connect in chains, rings, branches, and three-dimensional frameworks.
Because of this bonding ability, carbon creates millions of compounds with different properties. A small structural change can dramatically alter how a molecule behaves. For example, two molecules with the same atoms arranged differently may have completely different biological effects.
This structural diversity is why Organic Chemistry drives both life and industry. Carbohydrates provide energy, proteins support biological functions, and lipids build cell membranes. Meanwhile, synthetic organic compounds help create everything from stain-resistant fabrics to life-saving drugs.
Students who understand molecular structure gain a practical lens for predicting chemical behavior. This skill is valuable not only in exams but also in laboratory work and research careers.
Core Concepts Every Student Should Know
A strong Organic Chemistry foundation starts with a few essential ideas.
Functional Groups
Functional groups are specific arrangements of atoms that determine a molecule’s reactivity. Common examples include alcohols, aldehydes, ketones, carboxylic acids, amines, and esters.
Learning functional groups helps students quickly identify chemical behavior. For example:
Alcohols often participate in oxidation reactions
Carboxylic acids are acidic due to proton donation
Amines commonly act as bases
Instead of viewing molecules as random structures, students can break them into recognizable patterns.
Isomerism
Organic compounds often exist as isomers, molecules with the same molecular formula but different arrangements.
Structural isomers differ in connectivity, while stereoisomers differ in spatial arrangement. This distinction matters greatly in biology and medicine.
A famous example is drug chirality. One enantiomer of a compound may be therapeutic, while its mirror image may be inactive or harmful. Organic Chemistry helps students understand why molecular orientation matters.
Reaction Mechanisms
Reaction mechanisms explain how molecules transform step by step.
Rather than memorizing reactions, students benefit from learning electron movement. Curved arrows, nucleophiles, electrophiles, and intermediates tell the story of how bonds break and form.
Common reaction categories include:
Substitution
Addition
Elimination
Oxidation-reduction
Mechanistic thinking builds analytical skills useful in both academics and research.
Organic Chemistry in Product Innovation
Organic Chemistry is not limited to classrooms and laboratories. It directly influences the products people use daily.
Pharmaceuticals
Drug development depends heavily on organic synthesis. Chemists design molecules that interact with biological targets such as enzymes or receptors.
Pain relievers, antibiotics, antiviral treatments, and cancer therapies all emerge from Organic Chemistry research. Small structural modifications often improve drug effectiveness, reduce toxicity, or increase absorption.
For students interested in healthcare or medicinal chemistry, this field offers a direct link between molecular science and patient outcomes.
Sustainable Materials
Innovation in sustainable materials is another major application.
Biodegradable plastics, plant-based polymers, and recyclable coatings are created through organic design principles. As industries work to reduce waste and environmental harm, Organic Chemistry supports greener manufacturing.
Modern products increasingly prioritize sustainability, creating demand for chemists who understand both performance and environmental impact.
Consumer Products
Cosmetics, fragrances, detergents, food preservatives, and packaging materials all depend on organic compounds.
The texture of a skincare product, the scent of a perfume, and the shelf life of packaged food are influenced by molecular interactions studied in Organic Chemistry.
This makes the subject highly relevant even outside traditional laboratory careers.
Practical Advice for Students Learning Organic Chemistry
Many students struggle because they approach Organic Chemistry as a memorization-heavy subject. A more effective strategy is to treat it like a language with rules and recurring patterns.
Focus on Understanding, Not Memorization
Memorizing hundreds of reactions is inefficient. Instead, learn why reactions happen.
Ask questions such as:
Where are the electrons moving?
Which atom is electron-rich or electron-poor?
What makes this intermediate stable or unstable?
This mindset reduces cognitive overload.
Practice Drawing Structures Repeatedly
Organic Chemistry is visual. Drawing molecules by hand improves familiarity with bonding, stereochemistry, and reaction flow.
Practice converting between:
Lewis structures
Condensed formulas
Skeletal structures
Newman projections
The more often students draw, the faster recognition becomes.
Build a Functional Group Reference Sheet
Creating a personal summary sheet of functional groups, reactions, and properties can be highly useful.
Instead of relying on large textbooks during revision, students can review concise patterns and examples.
Solve Problems Consistently
Organic Chemistry improves through repetition.
Working through reaction problems, mechanism exercises, and synthesis challenges trains pattern recognition over time. Even 20 minutes of daily practice can be more effective than long study sessions before exams.
The Future of Organic Chemistry
The future of Organic Chemistry is closely tied to innovation.
Emerging areas include:
Drug discovery powered by AI
Renewable energy materials
Advanced polymers
Green catalysis
Precision agriculture chemicals
As science becomes increasingly interdisciplinary, Organic Chemistry remains foundational.
Students who master Organic Chemistry are not simply learning reactions. They are gaining tools to understand life at a molecular level and contribute to technologies that improve health, sustainability, and manufacturing.
Final Thoughts
Organic Chemistry is often called the chemistry of life, and for good reason. It explains the molecules behind biology, medicine, materials, and countless commercial products.
For students, the subject may initially appear demanding, but its logic becomes clearer with structured practice and conceptual understanding. By focusing on molecular patterns, reaction behavior, and real-world applications, learners can move beyond memorization and develop genuine scientific insight.
In both academia and industry, Organic Chemistry continues to shape innovation. Whether your future lies in medicine, research, environmental science, or product development, mastering Organic Chemistry opens the door to understanding and building the world at the molecular scale.
Publish by: belirangkalisari.id
0 comments
Log in to leave a comment.
Be the first to comment.