8 Insights Into Why Deuteration Technology Is Gaining Momentum in R and D Labs
Why Deuteration Technology Is Gaining Momentum in R&D Labs
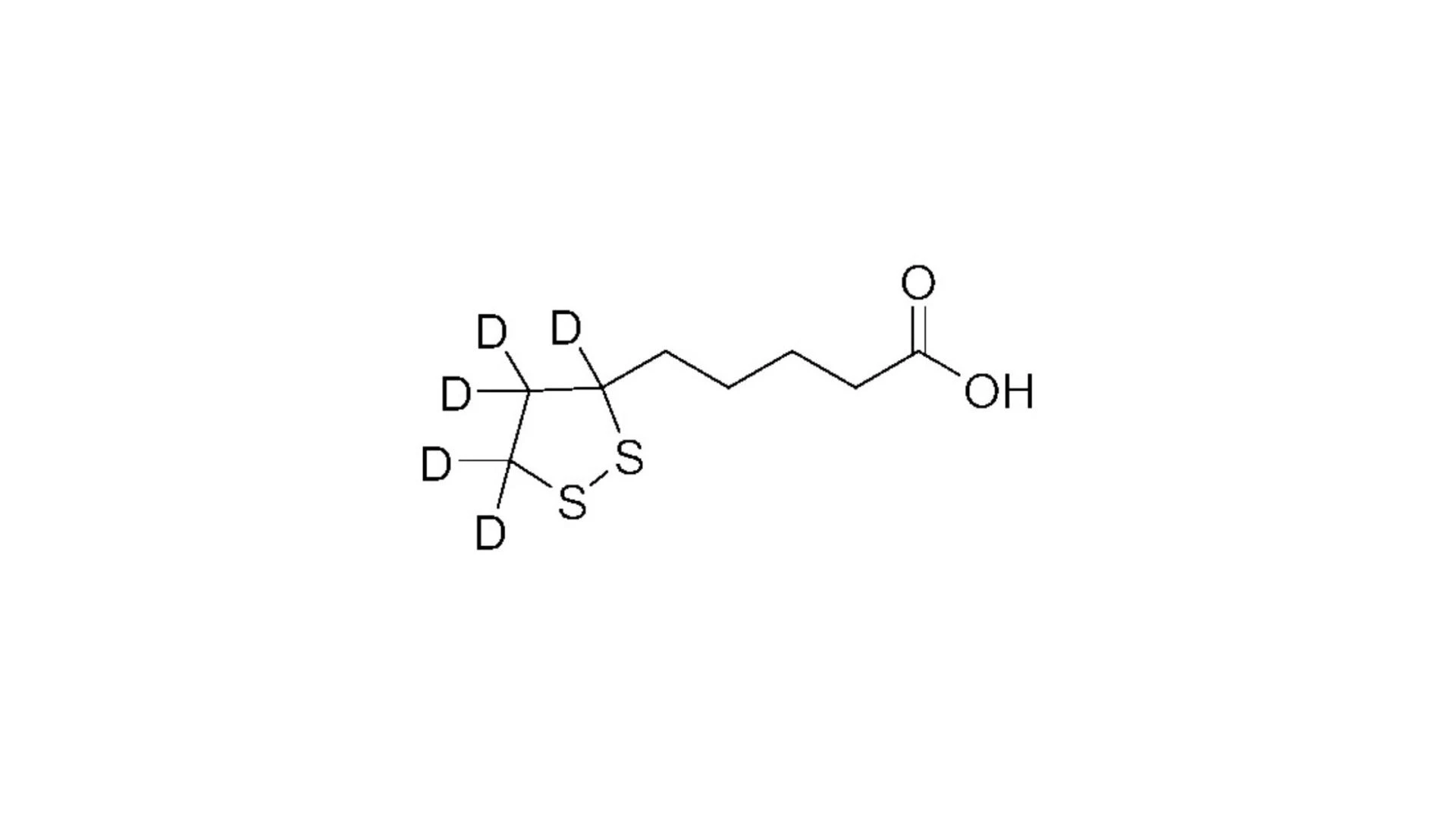
Drug candidates often fail not because they lack efficacy, but because metabolic instability, poor pharmacokinetics, or inconsistent performance limit their progression. Research teams are under pressure to refine molecules without restarting development from the beginning. This is where Deuteration Technology has gained serious scientific attention. By strategically replacing hydrogen with deuterium, molecular behavior can be improved without altering core pharmacology. For organizations focused on pharmaceutical product development, this approach offers a controlled and rational path toward better stability and measurable performance gains.
In competitive pipelines, incremental molecular improvements can determine whether a compound advances or stalls. Integrating Deuteration Technology within pharmaceutical product development strategies reflects a shift toward precision driven optimization rather than trial and error modification.
The Development Challenges Behind Promising Molecules
Even strong candidates encounter obstacles. Rapid metabolic breakdown reduces half life. Reactive metabolites introduce safety concerns. Reformulation attempts add cost and delay.
R and D leaders face difficult decisions. Should the molecule be redesigned. Should dosing be adjusted. Should additional studies be initiated. Each step consumes resources and introduces uncertainty.
Teams require solutions that preserve the therapeutic mechanism while improving metabolic profile and stability. This is why deuterium incorporation has become a practical tool rather than an academic concept.
How SV ChemBioTech Approaches Molecular Refinement
SV ChemBioTech applies deep synthetic expertise to support advanced isotopic labeling and structural modification. The company’s scientific foundation in complex organic synthesis enables precise hydrogen to deuterium substitution without compromising structural integrity.
Targeted Deuterium Incorporation
Strategic placement is essential. The focus is on metabolically vulnerable positions within the molecule, ensuring that modification delivers functional benefit rather than cosmetic change.
Strong Synthetic Control
Complex reaction pathways are carefully optimized to maintain yield and purity. Deuterated compounds require accuracy in execution to ensure reproducibility and analytical consistency.
Analytical Verification
Robust characterization confirms isotopic incorporation and structural accuracy. This ensures that research teams receive material that meets defined specifications.
Alignment With Development Goals
Rather than treating deuteration as a standalone service, SV ChemBioTech integrates it into broader development strategy, supporting stability studies and performance evaluation.
Through this approach, Deuteration Technology becomes a calculated enhancement tool within larger pharmaceutical product development frameworks.
The Science Behind the Momentum
Deuterium forms a stronger carbon deuterium bond compared to carbon hydrogen. This subtle change can slow metabolic oxidation at targeted sites. The result may include extended half life, reduced formation of unwanted metabolites, and improved pharmacokinetic stability.
The process begins with identifying metabolic hotspots. Synthetic pathways are then designed to introduce deuterium selectively. Reaction conditions are optimized to preserve molecular structure while achieving efficient incorporation. Analytical confirmation validates isotopic placement and purity.
For research teams, this systematic progression offers a data driven pathway toward measurable improvement without altering the therapeutic target.
Why R and D Teams Continue to Explore This Approach
Scientific teams value strategies that reduce redevelopment risk. Deuterium substitution allows optimization while maintaining the original pharmacological intent.
Collaborations with SV ChemBioTech often reflect a shared commitment to precision and disciplined execution. Clients appreciate clear communication, technical transparency, and reproducible results. Over time, consistent performance builds confidence in incorporating isotopic refinement into broader research plans.
Addressing Common Concerns
Some organizations question whether isotopic modification introduces regulatory complexity. Others wonder whether synthetic feasibility will limit scalability.
With experienced synthetic chemists and structured analytical validation, these concerns can be managed effectively. Careful route design supports reproducibility, while documentation practices maintain clarity throughout development stages.
Strengthening the Path Forward
As molecular design becomes increasingly sophisticated, incremental improvements can determine commercial viability. Deuteration Technology offers a rational method to enhance stability while preserving therapeutic intent. Within disciplined pharmaceutical product development programs, it provides measurable scientific advantage rather than speculative modification.
If your team is evaluating molecular refinement strategies, connect with SV ChemBioTech to explore how targeted isotopic incorporation can support your next stage of development with precision and confidence.
0 comments
Log in to leave a comment.
Be the first to comment.